Introduction
The status of lymph nodes is an independent prognostic factor in women with breast cancer [1, 2]. A sentinel node biopsy procedure is now considered as the standard of care for patients with early-stage breast cancer. In the Holycross Cancer Centre in Kielce, Poland the sentinel lymph node biopsy (SLNB) has been performed since 2007 [3].
Recently the treatment of cancer patients, particularly those with breast cancer, has become more patient-oriented and is strictly connected with the biological subtype of cancer [4–6]. But in clinically node-negative patients positive sentinel nodes are found and a decision about management in the axilla region is difficult, particularly in patients after a subcutaneous mastectomy and with a positive sentinel lymph node. Statistically in the majority of patients the Luminal A subtype is determined and the upfront therapy for these patients is surgery. Thus for positive sentinel node patients a proper strategy of treatment should be established.
The purpose of the study was to determine the correlation between biological subtype of breast cancer and the risk of its metastasis to a sentinel lymph node (SLN). The factors known prior to surgical treatment, namely age, tumour diameter, grade, type of cancer, and biological subtype, were evaluated.
Material and methods
In the analysed group there were 1018 consecutive patients with breast cancer, clinically node negative, untreated previously, in whom a sentinel lymph node biopsy procedure was performed. In 802 women a breast-conserving treatment, in 149 subcutaneous mastectomy and in 67 patients simple mastectomy were performed. All the patients were diagnosed and treated in the Holycross Cancer Centre.
Statistical analysis
Categorical variables were described with absolute frequencies and percentages and a χ2 test was used to compare categorical characteristics. Continuous variables were described by mean and standard deviations, the median and quartiles, as well as by minimum and maximum values. The normality of the distribution of the studied features was tested using the Shapiro-Wilk test. For features with normal distribution the t-test (for 2 groups) and the ANOVA test (for more than two groups) were used, for non-normal features the non-parametric tests of Mann-Whitney (for 2 groups) and Kruskal-Wallis (for more than two groups) were used. A receiver operating characteristic (ROC) curve analysis was applied. Sensitivity and specificity, positive and negative predictive values and confidence intervals were determined. The comparison of ROC curves was made. To identify the predictors of potential metastasis to the lymph nodes, univariate and multivariate logistic regression analyses were performed. A p-value < 0.05 was considered statistically significant. All statistical analysis was performed with the MedCalc Statistical Software version 18.10.2 (MedCalc Software bvba, Ostend, Belgium; http://www.medcalc.org; 2018).
Results
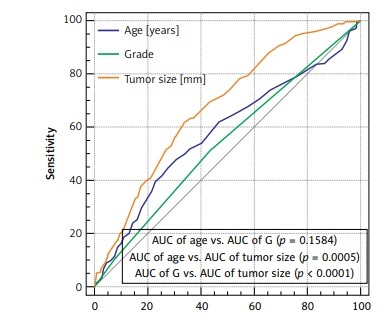
The median age was sixty-one. In 85% of cases an invasive carcinoma of no special type (NST) was diagnosed. In approximately 56% of cases the diameter of the tumour was no more than 20 mm. Luminal A subtype was recognised in 57% of patients. A positive sentinel lymph node was detected in 26.5% of women (Table I). The average number of removed sentinel nodes was 2.3 (median: 2, range 1–14). the average number of positive sentinel nodes was 1.4 (median: 1, range 1–8). The biological subtype was not a statistically significant factor in correlation with the probability of metastasis to a sentinel node (p = 0.4028) (Table II). Age was not statistically significant in relation to the entire group, as well as for patients with or without metastases to the sentinel node (p = 0.0697). However, patients with Luminal A and positive SLN were younger; the median age was 57 years vs. 63 years for Luminal A negative SLN (p = 0.0001). In Luminal B patients with SLN metastasis the age was 6.5 years lower than in the SLN negative ones (p = 0.0248) (Table III). Tumour size was statistically significant in relation to biological subtype and sentinel node status. In triple negative and non-luminal subtypes this correlation was not found (p = 0.5953 and p = 0.4248 respectively) (Table IV). A statistically significant association was found for biological subtype and tumour grade (p < 0.0001) (Table V). A receiver operating characteristic analysis was performed for sentinel node negative and positive status and for independent variables: age, grade and tumour size. Outcomes are shown in Figure 1. Sensitivity, specificity, +PV (positive predictive value), –PV (negative predictive value) for age were respectively: 44.8%, 72.3%, 36.6%, 78.6%; for grade respectively: 51.5%, 56.1%, 29.6%, 75.8%; and for tumour size respectively: 61.9%, 66.1%, 39.5%, 82.9%. Analysed features show the greatest sensitivity and specificity for tumour size. At the same time, all of them have high –PV. All of them are statistically significant. The biggest area under the curve (AUC) was estimated for tumour size and it is statistically significantly different from AUC for age (p = 0.0005) and tumour grade (p < 0.0001). A significant difference for AUC for age and tumour grade was not found (p = 0.1584) (Figure 1). Table VI shows outcomes of the univariate analysis for biological subtypes and for the entire group. The following proved to be statistically significant: for Luminal A, age (p = 0.0001), tumour size (p = 0.0007), tumour grade (p = 0.0259) and type of cancer (p = 0.0326); for Luminal B, age (p = 0.0039) and tumour size (p = 0.0284); for Luminal B HER2 positive, tumour size only (p = 0.0162); for the entire group, age (p = 0.0001), tumour size (p < 0.0001), and tumour grade (p = 0.0228). For factors which were statistically significant in the univariate analysis, multivariate analysis was performed. Among them only age and tumour size proved to be significant for the entire group, respectively: OR = 0.59, p = 0.0004; OR = 1.96; p < 0.0001; for the Luminal A subtype, OR = 0.51, p = 0.0007; OR = 1.78, p = 0.0045, respectively. For Luminal B patients, in women over 61 years, the risk of sentinel node metastasis probability decreases by 67% and for tumours over 21 mm the probability of a positive sentinel node metastasis increases by 117% (Table VII).
Table I
Characteristics of the entire group of patients
| Factor | Number |
|---|---|
| N | 1018 |
| Age: | |
| Mean (SD) | 60 (11) |
| Median (Q1–Q2) | 61 (52–67) |
| Min.–max. | 28–92 |
| Age [years]: | |
| ≤ 61 | 690 (67.8%) |
| > 61 | 328 (32.2%) |
| Side: | |
| Left | 518 (50.9%) |
| Right | 500 (49.1%) |
| Type of cancer: | |
| No specific type | 870 (85.5%) |
| Lobular cancer | 98 (9.6%) |
| Other* | 50 (4.9%) |
| Grade: | |
| 1 | 550 (54.1%) |
| 2 | 382 (37.6%) |
| 3 | 85 (8.4%) |
| Tumour size: | |
| Mean (SD) | 20.6 (10.8) |
| Median (Q1–Q2) | 20 (13–26) |
| Min.–max. | 1–80 |
| Tumour size [mm]: | |
| ≤ 21 | 429 (41.1%) |
| > 21 | 589 (57.9%) |
| pT: | |
| 1 | 572 (56.2%) |
| 2 | 432 (42.4%) |
| 3 | 13 (1.3%) |
| 4 | 1 (0.1%) |
| Sentinel node status: | |
| N negative | 748 (73.5%) |
| N positive | 270 (26.5%) |
| Biological subtype: | |
| Luminal A | 582 (57.2%) |
| Luminal B | 160 (15.7%) |
| Luminal B progesterone negative receptor | 102 (10.0%) |
| Luminal B HER2 positive | 77 (7.6%) |
| Triple negative | 74 (7.3%) |
| Non luminal | 23 (2.3%) |
Table II
Relationship between sentinel node status and biological subtype
Table III
Relationship between age and biological subtype and sentinel node status
Table IV
Relationship between tumour size and biological subtype and sentinel node status
Table V
Relationship between tumour grade and biological subtype
Table VI
Univariate analysis of risk factors of sentinel lymph node metastasis
Table VII
Multivariate analysis of risk factors of sentinel node metastasis (ENTER method)
Discussion
Luminal A tumours are the most common breast cancers. Other biological subtypes are less numerous [7–10]. Additionally in our series, Luminal B cancer subtype with negative expression of a progesterone receptor was detected, which is associated with different biology and prognosis in this particular subtype [11–13]. A sentinel lymph node biopsy can be performed in every patient with any biological subtype of breast cancer, even in triple negative and epidermal growth factor receptor 2 (HER2) positive patients in whom the strategy of treatment has recently changed [14–17]. Systemic and antiHER2 therapy is applied in these patients at the beginning of the therapy, excluding patients in whom the tumour diameter is less than 10 mm. Even in early stage disease we decide on neoadjuvant chemo- and antiHER2 therapy. In many cases regression is achieved and non-harmful axillary surgery is used, replaced by a sentinel node biopsy procedure.
The available published data indicate that the risk of regional lymph node metastasis in triple negative breast cancer patients is low and an SLNB procedure may be not necessary [17–19]. However, still, for the aforementioned patients, even after a neoadjuvant systemic therapy, an SLNB procedure should be performed. In 40% of triple negative and HER2 positive cancers pathological complete regression has been achieved (own data, not published); thus this particular form of treatment should be applied.
Carcinogenesis of breast cancer is different in older women, and its development can last years, in contrast to young women. The most common subtype is Luminal A. Luminal A breast cancers are usually of low histological grade, with slow growth and a good prognosis, but quite frequently in young women the illness is more advanced at the time of making a diagnosis [20, 21].
Statistically, in the analysed group, the probability of sentinel lymph node metastasis did not differ between particular subtypes of cancers. For Luminal A and Luminal B cancers the following factors were proven to be statistically significant in relation to the risk of SLN metastasis: age, grade and tumour size, which is consistent with data reported in the literature [4, 10, 18, 22, 23]. Other authors point to different factors influencing the presence of metastasis in sentinel nodes. Majid et al. found that the presence of tumours measuring > 20 mm, multifocality and lymphovascular invasion are strong predictive factors for SLN metastases [24]. According to a study by Ding et al. involving 417 patients, a univariate analysis revealed age, tumour size, histological grade and Ki-67 index to be associated with SLN metastases, while Ki-67 index was not found to be significant in a multivariate analysis [10]. In a series of 324 patients, Mao found no association between Ki-67 index and SLN status lower or higher than 14. A Ki-67 index > 14 percent has been reported to be associated with SLN metastases [25–28]. Other investigators indicate a relationship between tumour-infiltrating lymphocytes (TILs) and higher probability of regional lymph nodes metastases. Macchetti et al. discovered a statistically significant correlation between CD4-positive TILs and axillary lymph node involvement, but did not find a correlation between CD8-positive TILs and axillary lymph node involvement [29]. Conversely, Matkowski et al. found a highly significant correlation between both CD4-positive and CD8-positive TILs and axillary lymph node involvement [30]. According to our own data a precise radiological evaluation should be performed in women under sixty and in highly malignant tumours of over 20 mm in diameter. A very thorough diagnostic work-up should be performed not only in relation to a primary tumour but also to regional lymph nodes. A further strategy after surgery for Luminal A cancer patients is connected with the number of positive sentinel lymph nodes. Even a macrometastasis to a sentinel node in Luminal A cancer patients is not an indication for axillary lymphadenectomy, which can be replaced by a radiotherapy of regional lymph nodes. In sentinel node positive patients, no additional metastases are found in the remaining lymph nodes removed on axillary lymphadenectomy in about 50–60% of patients [31]. Subsequently in several countries axillary radiotherapy is approved in lieu of axillary lymphadenectomy [32–34]. Supplementary chemotherapy can be applied in the case of numerous positive sentinel lymph nodes. In the case of existence of metastases to one or two sentinel lymph nodes usually chemotherapy is not provided for these patients and hormone therapy is the treatment of choice regarding systemic treatment. Conversely, in Luminal B, Luminal B HER 2 positive, triple negative or non-luminal patients, chemotherapy and anti-HER2 therapy are used [17, 35].
In concluion, according to our analysis, luminal breast cancers are the most numerous subtypes and in these cases we expect more frequent instances of metastasis to a sentinel node. Based on our outcomes we can state that the risk of a sentinel node metastasis decreases in women older than 61 years for Luminal A by 54%, Luminal B by 64%, and for the entire group by 46%. A tumour size bigger than 21 mm increases the probability of a positive sentinel node for Luminal A and Luminal B by 93% and 150%, respectively. The increase of the tumour grade causes the higher probability of a sentinel node metastasis, for Luminal A by 50%, for the entire group by 28%. Following the most up-to-date and modern procedures of breast cancer patients’ treatment, a procedure of a sentinel lymph node biopsy is used, replacing an aggressive treatment in the axilla region.