Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
NEUROSURGERY / BASIC RESEARCH
Long noncoding RNA RP11-704M14.1 acts as a sponge of miR-6756-5p to promote neuronal proliferation by regulating ALOX15 expression
1
Brain Injury Centre, Department of Neurosurgery, Renji Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
2
Shanghai Institute of Head Trauma, Shanghai, China
3
Department of Neurosurgery, Third Affiliated Hospital of Naval Medical University, Shanghai, China
4
Department of Neurosurgery, Shanghai Pudong New Area People’s Hospital, Shanghai, China
These authors had equal contribution to this work
Submission date: 2024-03-11
Final revision date: 2024-04-30
Acceptance date: 2024-05-15
Online publication date: 2024-06-12
Corresponding author
Zheng Ping
Shanghai Pudong New Area People\'s Hospital, 490, South Chuanhuan Road, 201200, Shanghai, China
Shanghai Pudong New Area People\'s Hospital, 490, South Chuanhuan Road, 201200, Shanghai, China
KEYWORDS
competing endogenous RNAlong noncoding RNARP11-704M14.1single-cell RNA sequencingtraumatic brain injury
TOPICS
ABSTRACT
Introduction:
Long noncoding RNAs (lncRNAs) mediate critical effects in central nervous system diseases; however, the exact role of lncRNAs in human traumatic brain injury (TBI) remains elusive. Considering the principles of primary prediction, targeted prevention, and personalized treatment medicine (PPPM), identifying specific novel biomarker associated with TBI and exploring the underlying mechanisms comprehensively are crucial steps towards achieving primary prediction, targeted prevention, and personalized treatment of TBI.
Material and methods:
In this study, we integrated single-cell RNA sequencing (sc-RNA-seq) data and microarray chipset data for TBI to identify a competing endogenous RNA axis.
Results:
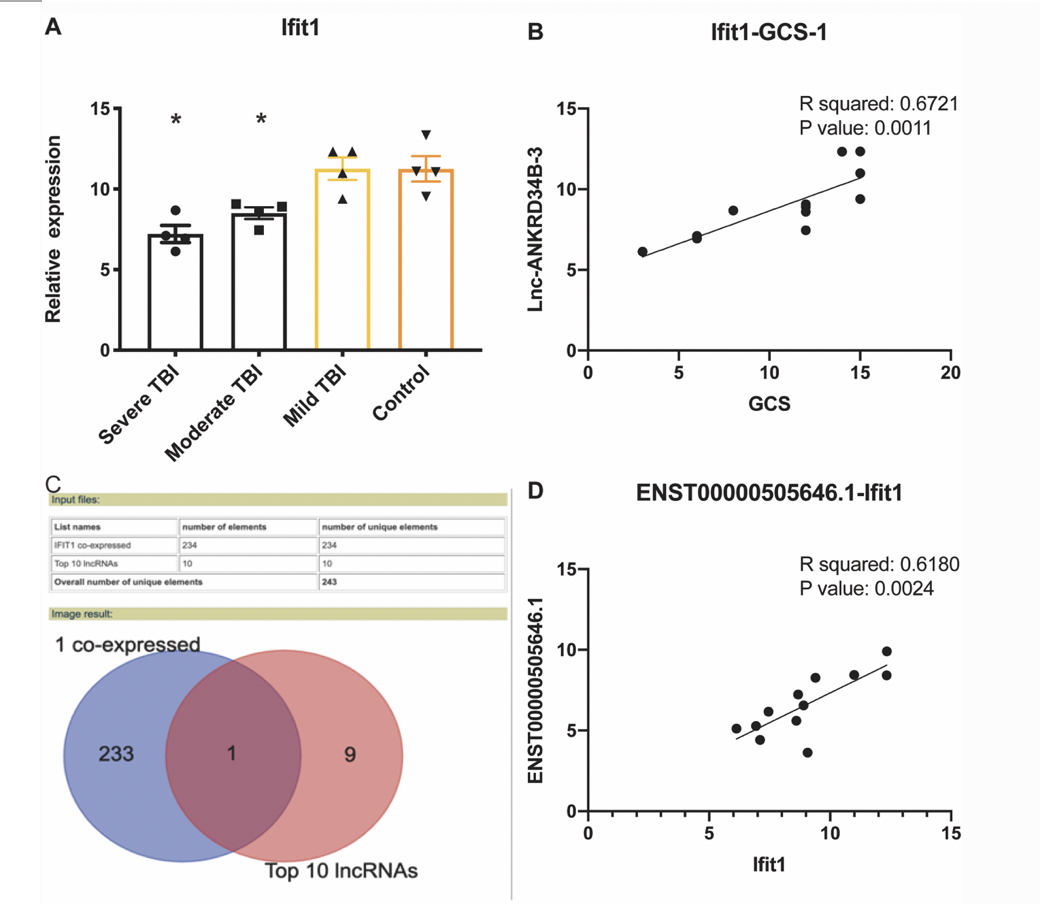
We detected 11 clusters based on the sc-RNA-seq data and identified Ifit1 as a marker gene co-expressed with ENST00000505646.1 (RP11-704M14.1). Additionally, the expression of IFIT1 and RP11-704M14.1 was found to decrease with the severity of TBI. RP11-704M14.1 promoted neuronal proliferation, and its knockdown prevented this effect. Furthermore, we found that RP11-704M14.1 functions as a miR-6756-5p sponge and increases the expression of its target gene ALOX15.
Conclusions:
Our results show that RP11-704M14.1 promotes neuronal proliferation by sponging miR-6756-5p and regulating ALOX15 expression in brain insults; accordingly, targeting this lncRNA presents a promising avenue for advancing the transition from reactive medicine to PPPM in managing traumatic brain injury, potentially leading to significant clinical benefits.
Long noncoding RNAs (lncRNAs) mediate critical effects in central nervous system diseases; however, the exact role of lncRNAs in human traumatic brain injury (TBI) remains elusive. Considering the principles of primary prediction, targeted prevention, and personalized treatment medicine (PPPM), identifying specific novel biomarker associated with TBI and exploring the underlying mechanisms comprehensively are crucial steps towards achieving primary prediction, targeted prevention, and personalized treatment of TBI.
Material and methods:
In this study, we integrated single-cell RNA sequencing (sc-RNA-seq) data and microarray chipset data for TBI to identify a competing endogenous RNA axis.
Results:
We detected 11 clusters based on the sc-RNA-seq data and identified Ifit1 as a marker gene co-expressed with ENST00000505646.1 (RP11-704M14.1). Additionally, the expression of IFIT1 and RP11-704M14.1 was found to decrease with the severity of TBI. RP11-704M14.1 promoted neuronal proliferation, and its knockdown prevented this effect. Furthermore, we found that RP11-704M14.1 functions as a miR-6756-5p sponge and increases the expression of its target gene ALOX15.
Conclusions:
Our results show that RP11-704M14.1 promotes neuronal proliferation by sponging miR-6756-5p and regulating ALOX15 expression in brain insults; accordingly, targeting this lncRNA presents a promising avenue for advancing the transition from reactive medicine to PPPM in managing traumatic brain injury, potentially leading to significant clinical benefits.
REFERENCES (41)
1.
GBD 2016 Traumatic Brain Injury and Spinal Cord Injury Collaborators. Global, regional, and national burden of traumatic brain injury and spinal cord injury, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurology 2019; 18: 56-87.
2.
Yin X, Shen L, Zhou L, et al. Duodenal versus gastric feeding in patients with traumatic brain injury: a systematic review and meta-analysis. Arch Med Sci 2022; https://doi.org/10.5114/aoms/1....
3.
Lu S, Yang X, Wang C, et al. Current status and potential role of circular RNAs in neurological disorders. J Neurochem 2019; 150: 237-48.
4.
Zhang L, Wang H. Long non-coding RNA in CNS injuries: a new target for therapeutic intervention. Mol Ther Nucleic Acids 2019; 17: 754-66.
5.
Shan W, Chen W, Zhao X, et al. Long noncoding RNA TUG1 contributes to cerebral ischaemia/reperfusion injury by sponging mir-145 to up-regulate AQP4 expression. J Cell Mol Med 2020; 24: 250-9.
6.
Zhang X, Liu Z, Shu Q, et al. LncRNA SNHG6 functions as a ceRNA to regulate neuronal cell apoptosis by modulating miR-181c-5p/BIM signalling in ischaemic stroke. J Cell Mol Med 2019; 23: 6120-30.
7.
Wang CF, Zhao CC, Weng WJ, et al. Alteration in long non-coding RNA expression after traumatic brain injury in rats. J Neurotrauma 2017; 34: 2100-8.
8.
Li Z, Han K, Zhang D, et al. The role of long noncoding RNA in traumatic brain injury. Neuropsychiatr Dis Treat 2019; 15: 1671-7.
9.
Patel NA, Moss LD, Lee JY, et al. Long noncoding RNA MALAT1 in exosomes drives regenerative function and modulates inflammation-linked networks following traumatic brain injury. J Neuroinflamm 2018; 15: 204-23.
10.
Yang L, Xu Y, Zhang W. Sophoricoside attenuates neuronal injury and altered cognitive function by regulating the LTR-4/NF-B/PI3K signalling pathway in anaesthetic-exposed neonatal rats. Arch Med Sci 2020; 20: 248-54.
11.
Jiang YJ, Cao SQ, Gao LB, et al. Circular ribonucleic acid expression profile in mouse cortex after traumatic brain injury. J Neurotrauma 2019; 36: 1018-28.
12.
Huang S, Ge X, Yu J, et al. Increased miR-124-3p in microglial exosomes following traumatic brain injury inhibits neuronal inflammation and contributes to neurite outgrowth via their transfer into neurons. FASEB J 2018; 32: 512-28.
13.
Wu J, He J, Tian X, et al. microRNA-9-5p alleviates blood-brain barrier damage and neuroinflammation after traumatic brain injury. J Neurochem 2020; 153: 710-26.
14.
Ding Y, Zhu W, Kong W, et al. Edaravone attenuates neuronal apoptosis in hippocampus of rat traumatic brain injury model via activation of BDNF/TrkB signaling pathway. Arch Med Sci 2021; 17: 514-22.
15.
He B, Chen W, Zeng J, et al. Long noncoding RNA NKILA transferred by astrocyte-derived extracellular vesicles protects against neuronal injury by upregulating NLRX1 through binding to mir-195 in traumatic brain injury. Aging 2021; 13: 8127-45.
16.
Tao Z, Guo H, Tang J, Cheng M. Knockdown of SNW1 ameliorates brain microvascular endothelial cells injury by inhibiting NLRP3 inflammasome activation. Arch Med Sci 2021; https://doi.org/10.5114/aoms/1....
17.
Zheng P, Shu L, Ren D, et al. circHtra1/miR-3960/GRB10 axis promotes neuronal loss and immune deficiency in traumatic brain injury. Oxid Med Cell Longev 2022; 2022: 3522492.
18.
Ren D, Chen W, Cao K, et al. Expression profiles of long non-coding RNA and messenger RNA in human traumatic brain injury. Mol Ther Nucleic Acids 2020; 22: 99-113.
19.
Witcher KG, Bray CE, Chunchai T, et al. Traumatic brain injury causes chronic cortical inflammation and neuronal dysfunction mediated by microglia. J Neurosci 2021; 41: 1597-616.
20.
Hao Y, Hao S, Andersen-Nissen E, et al. Integrated analysis of multimodal single-cell data. Biorxiv 2020; 2020.10.12.335331.
21.
Aran D, Looney AP, Liu L, et al. Reference-based analysis of lung single-cell sequencing reveals a transitional profibrotic macrophage. Nat Immunol 2019; 20: 163-72.
22.
Jin S, Guerrero-Juarez CF, Zhang L, et al. Inference and analysis of cell-cell communication using CellChat. Nat Commun 2021; 12: 1088.
23.
Cao J, Spielmann M, Qiu X, et al. The single-cell transcriptional landscape of mammalian organogenesis. Nature 2019; 566: 496-502.
24.
Chen W, He B, Tong W, et al. Astrocytic insulin-like growth factor-1 protects neurons against excitotoxicity. Front Cell Neurosci 2019; 13: 298.
25.
He B, Chen W, Zeng J, et al. MicroRNA-326 decreases tau phosphorylation and neuron apoptosis through inhibition of the JNK signaling pathway by targeting VAV1 in Alzheimer’s disease. J Cell Physiol 2020; 235: 480-93.
26.
Agarwal V, Bell GW, Nam JW, Bartel DP. Predicting effective microRNA target sites in mammalian mRNAs. Elife 2015; 4: e05005.
27.
Chen Y, Wang X. miRDB: an online database for prediction of functional microRNA targets. Nucleic Acids Res 2019; 48: D127-131.
28.
Backes C, Fehlmann T, Kern F, et al. miRCarta: a central repository for collecting miRNA candidates. Nucleic Acids Res 2017; 46: gkx851.
29.
Wang S. Investigation of long non-coding RNA expression profiles in patients with post-menopausal osteoporosis by RNA sequencing. Exp Ther Med 2020; 20: 1487-97.
30.
Lim CS, Porter DW, Orandle MS, et al. Resolution of pulmonary inflammation induced by carbon nanotubes and fullerenes in mice: role of macrophage polarization. Front Immunol 2020; 11: 1186.
31.
Rohwer N, Chiu C, Huang D, et al. Omega-3 fatty acids protect from colitis via an Alox15-derived eicosanoid. FASEB J 2021; 35: e21491.
32.
Kroschwald S, Chiu CY, Heydeck D, et al. Female mice carrying a defective Alox15 gene are protected from experimental colitis via sustained maintenance of the intestinal epithelial barrier function. Biochim Biophys Acta Mol Cell Biol Lipids 2018; 1863: 866-80.
33.
Zhang P, Xing X, Hu C, et al. 15-Lipoxygenase-1 is involved in the effects of atorvastatin on endothelial dysfunction. Mediat Inflamm 2016; 2016: 6769032.
34.
Shalini SM, Ho CFY, Ng YK, et al. Distribution of Alox15 in the rat brain and its role in prefrontal cortical resolvin D1 formation and spatial working memory. Mol Neurobiol 2018; 55: 1537-50.
35.
Kwon HJ, Kim SN, Kim YA, Lee YH. The contribution of arachidonate 15-lipoxygenase in tissue macrophages to adipose tissue remodeling. Cell Death Dis 2016; 7: e2285.
36.
Chen Y, Peng C, Abraham SA, et al. Arachidonate 15-lipoxygenase is required for chronic myeloid leukemia stem cell survival. J Clin Invest 2014; 124: 3847-62.
37.
Anderson GD, Farin FM, Bammler TK, et al. The effect of progesterone dose on gene expression after traumatic brain injury. J Neurotraum 2011; 28: 1827-43.
38.
Wang L, Zhao J, Zhu B, et al. Microglia polarization in heat-induced early neural injury. Arch Med Sci 2024; 20: 1307-13.
39.
Pichlmair A, Lassnig C, Eberle CA, et al. IFIT1 is an antiviral protein that recognizes 5-triphosphate RNA. Nat Immunol 2011; 12: 624-30.
40.
Choi YJ, Bowman JW, Jung JU. A Talented duo: IFIT1 and IFIT3 patrol viral RNA caps. Immunity 2018; 48: 474-6.
41.
Kikkert M. Innate immune evasion by human respiratory RNA viruses. J Innate Immun 2020; 12: 4-20.