Introduction
Bladder cancer (BCa) is the second most malignant tumor of the urinary system in western society, but in China it is the most common urological malignancy, with an increased incidence [1, 2]. It is estimated that over 80,000 new cases of BCa and 17,000 cases of bladder cancer-related deaths will occur in America this year [3]. Overall, 70–80% of patients have been diagnosed with well-differentiated or moderately differentiated non-muscle invasive BCa. Despite clinical therapeutics, still 60–70% of patients suffered recurrence, of whom 10–30% eventually had malignant progression, including muscle invasiveness or metastasis [4–6]. Thus, it is necessary for clinicians to identify high-risk patients. Moreover, there is a need to study BCa progression mechanisms to develop better therapies, potential biomarkers, and reduce BCa-related morbidity and mortality.
The interleukin 1 (IL-1) cytokine family includes 3 major members that bind to the same receptor, including 2 isoforms of IL-1 (α and β)and the antagonist IL-1Ra [7–9]. Interleukin 1α (IL-1α) is a key cytokine involved in innate and adaptive immunity, inflammasome activation and hematopoiesis through the expression of metastatic and angiogenic genes and growth factors. Owing to the general characteristics IL-1α is associated with carcinogenesis and tumor progression. Recent research showed that IL-1, as a major pro-inflammatory (“alarm”) cytokine that affects both immunity and hematopoiesis, play important roles in tumor initiation, development, and metastasis [10–12]. However, to our knowledge, the biological role of IL-1α in BCa remains unclear.
In this study, we found that high levels of IL-1α expression were significantly associated with poor clinical outcome in BCa patients, and further functional assays showed that knockdown of IL-1α might inhibit the ability of cancer cells to proliferate and invade or migrate both in vivo and in vitro. Based on the findings, IL-1α may be a prognostic biomarker for predicting BCa malignant progression and serve as a novel target in cancer therapy.
Material and methods
Patients and samples
One hundred twenty-three patients (aged 54–69 years) clinically and pathologically diagnosed with urothelial carcinoma of bladder in our hospital between July 2013 and May 2017 were included in our study. Tumor tissues were obtained after the first surgical treatment. The clinical and pathological characters such as age, gender, pTMN stage, tumor grade, tumor sizes, lymph metastasis and vascular invasion were recorded. All applicable international, national, and/or institutional guidelines for the care and use of human specimens were followed. Patients were classified according to the 2009 Union Internationale Contre le Cancer Tumour Node Metastasis staging and the 2004 World Health Organization International Society of Urologic Pathologists classification. All patients had no preoperative radiotherapy and/or chemotherapy and neoadjuvant chemotherapy. Attendees provided written informed consent. All patients showed no evidence of tumor metastasis confirmed by cross-sectional imaging. The study was carried out in accordance with the guidelines approved by the Ethics Committee of Tianjin First Center Hospital.
Immunohistochemistry analysis
Immunohistochemical (IHC) staining was performed using Histostain – SP Kits (SPN-9001, ZSGB-BIO, China). Rabbit antibody against IL-1α (1 : 100 dilution, ab7632, Abcam, UK) was used following the manufacturer’s instructions. The method was described previously [13–15]. For the results, IL-1α cytoplasmic staining was scored using a 4-point scale (0, no staining; 1+, light staining at high magnification; 2+, intermediate staining; 3+, dark staining of linear membrane at low magnification). Additionally, an immunostaining score (H-score) was measured by the multiplication of IL-1α stained cells and the corresponding intensity score [13–15]. According to the distribution of H-scores, the IL-1α was divided into high and low expression groups.
Cell culture and reagents
T24 and 5637 cells were cultured in RPMI1640 (Invitrogen), supplemented with 10% heat-inactivated fetal bovine serum, 100 U/ml penicillin, and 0.1 mg/ml streptomycin in a 5% CO2 atm at 37°C.
Cell line generation
Small hairpin RNA (shRNA) virus (target virus and negative control) was generated from Vigene (Cat# SH833111, Vigene Biosciences Inc, Maryland, USA) for human IL-1α (RefSeq number: NM_000575) downregulation. Cells were transfected with virus according to the manufacturer’s instructions. Reverse transcription quantitative polymerase chain reaction (RT-q-PCR) and western blot were used to confirm the IL-1α silencing effect.
RNA analysis
Total RNA was extracted by the TRIzol reagent (Invitrogen). A Transcriptor Reverse Transcriptase kit (Roche) was used for first strand cDNA generation. RT-q-PCR was performed with SuperReal PreMix (Tiangen Biotech) by following the amplification scheme: 95°C for 2 min (1 cycle), 95°C for 5 s, 55°C for 30 s, and 72°C for 30 s (35 cycles) and final extension at 72°C for 6 min. A melting curve analysis was performed using the Applied Biosystems StepOne system to ensure correct DNA production. Using 2–ΔΔCt method, the relative expression level was normalized by the internal standard GAPDH. All experiments were performed in triplicate and repeated 3 times. Primer sequences were as follows:
Western blot analysis
Cells were washed by phosphate buffered saline (PBS) once and then suspended in lysis buffer (50 mM Tris-HCl, pH 7.40, 150 mM NaCl, 1 mM EDTA, 1% deoxycholic acid, 1% NP-40 and protease inhibitor cocktail). Protein concentrations were measured by the BCa Protein Assay (Thermo Fisher Scientific) according to the manufacturer’s instructions. Next, an equal level of protein was electrophoresed by sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred onto Immobilon-P transfer membrane (Merck Millipore). The antibodies were as follows: rabbit antibody against IL-1α (1 : 1000 dilution, ab7632, Abcam, UK), mouse antibody against β-actin (1 : 1000 dilution, ab8226, Abcam, UK), rabbit antibody against Ki67 (1 : 1000 dilution, ab16667, Abcam, UK), mouse antibody against proliferating cell nuclear antigen (PCNA) (1 : 500 dilution, ab29, Abcam, UK), mouse antibody against metalloproteinase (MMP)-2 (1 : 1000 dilution, ab37150, Abcam, UK), and mouse antibody against MMP-9 (1 : 1000 dilution, ab38898, Abcam, UK).
Colony formation assay
800 cells per well were plated onto 6-well plates. Cells were fixed with methyl hydrate, stained with 0.5% crystal violet solution and counted after 2 weeks. The assays were performed in triplicate and repeated 3 times.
MTT proliferation assay
MTT assay was performed to detect cell proliferation. 2 × 103 cells were inoculated in a 96-well plate, and 20 μl of 5 mg/ml MTT solution was added to each well 5 h before the end of the incubation. After dimethyl sulfoxide was added to the wells, the plate was transferred to a plate reader (Bio-Rad, Hercules) and absorbance at 570 nm wavelength was measured.
In vitro scratch assay
In vitro scratch assay was performed to study cell migration changes. Trypsinized cells were seeded in 6-well plates and grew to full confluence overnight. A sterile pipette tip was used to scrape the cell monolayer in a straight line to create a scratch in the middle of each well. Subsequently, the medium was changed and replaced with fresh complete medium. The area of the wound closed was calculated at indicated time points.
Migration and invasion assay
For migration or invasion assay, 5 × 104 cells were inoculated in the upper chamber with solid growth factor reduced Matrigel (BD Biosciences, Bedford, MA, USA). The complete medium was placed in the lower chamber and cells were allowed to migrate for 12 h at 37°C. Next, cells that had migrated through the filter pores from the underside of the membrane were fixed and stained. The number of cells in each chamber was counted under 3 randomized fields by microscope.
Xenograft study
The in vivo animal study was performed with approval by the Animal Experimentation Ethics Committee of Tianjin First Center Hospital. All immune-deficient nude mice (aged 4 weeks) were purchased from Beijing HFK bioscience Co., Ltd. (Beijing, China). 2 × 106 negative control shRNA treated T24 cells and 2 × 106 IL-1α targeting shRNA treated T24 cells were transplanted into mice to establish the xenograft tumor models. Thereafter, tumor final volume was recorded.
Statistical analysis
All data were proceeded by SPSS 22.0 software for Windows (SPSS Inc., Chicago, IL, USA). The clinical relationship between IL-1α expression level and BCa patients’ specific survival rate was evalated by the Kaplan-Meier curve method. Results were analyzed by independent Student’s t-test. Results and data are expressed as the mean ± standard deviation. P-value < 0.05 was defined as statistically significant.
Results
Increased IL-1α 6 expression in patients with bladder cancer correlated with poor clinical outcome
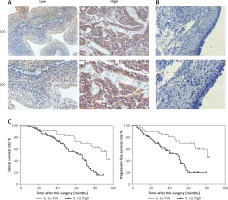
In our clinical cohort of 123 BCa patients, IHC analysis was used to detect low and high expression intensity of IL-1α in BCa specimens (Figure 1 A), while IL-1α was negative in the normal bladder tissues adjacent to carcinoma (Figure 1 B). Based on the protein expression level of IL-1α in the BCa patients, the cohort was divided into 2 groups: the IL-1α low expression group (n = 40) and IL-1α high expression group (n = 83) (Table I). In particular, patients with high IL-1α expression intensity were associated with malignant distant metastasis (p = 0.020) and vascular invasion (p = 0.025) compared with the low-intensity group. There was no clear clinical correlation between IL-1α expression and other clinicopathological features including age, gender, tumor stage, tumor grade, lymph node metastasis, and recurrence. To investigate the clinical relationship between IL-1α expression level and BCa patients’ specific overall survival rate and progression-free survival rate, the Kaplan-Meier curve method was performed. The data showed that the IL-1α high expression group tended to achieve a short overall survival rate and progression-free survival rate compared with the low-intensity group (p < 0.05, respectively, Figure 1 C). In summary, the results suggested that IL-1α was highly expressed in BCa tumors while highly expressed IL-1α was closely related to short overall survival rate and progression-free survival rate in BCa patients.
Table I
Relationships of interleukin 1α (IL-1α) and clinicopathological characteristics in 123 patients with bladder cancer
Figure 1
Increased interleukin 1α (IL-1α) expression associated with poor clinical outcome. A – Low and high IL-1α expression intensity in the bladder cancer tissues with immunohistochemistry (IHC) was observed. B – Low IL-1α expression intensity in the normal bladder tissues adjacent to carcinoma with IHC was observed. C – Overall survival rate and progression-free survival rate in our clinical cohort: high IL-1α protein expression level was significantly associated with lower overall survival rate and progression-free survival rate compared with low expression group (p < 0.05, respectively)
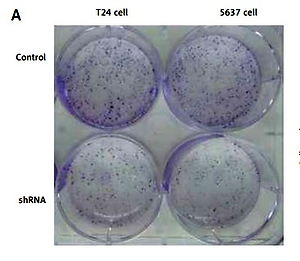
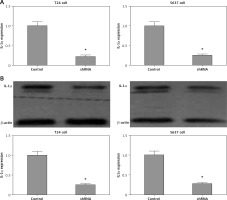
The adeno-associated virus based IL-1α stable knockdown cell lines construction
By recombinant lentivirus, IL-1α expression in cancer cells was silenced by transfection in T24 and 5637 cells. In contrast to negative control cells, we generated stable cell lines with significantly reduced IL-1α mRNA and protein levels validated by RT-q-PCR and Western blot, respectively (both p < 0.05, Figures 2 A and B). We could further efficiently investigate the IL-1α role in BCa by using this model.
Figure 2
mRNA and protein expression level of interleukin 1α (IL-1α) in stable cell lines. A – reverse transcription quantitative polymerase chain reaction assay. Results showed that IL-1a mRNA expression levels were significantly reduced compared to negative control cells (p < 0.05). B – Western blot. Results showed that IL-1α protein expression levels were significantly reduced compared to negative control cells (p < 0.05)
All results were obtained from at least 3 independent experiments.
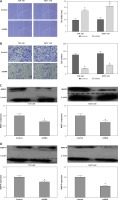
Knockdown of IL-1α reducing cell proliferation by modulating Ki-67 and proliferating cell nuclear antigen proteins in T24 and 5637 cell lines
To study the biological effects of IL-1α, colony formation assay and MTT proliferation assay were conducted to assess the possible proliferative effects in BCa cell lines. Colony formation assay results showed that the IL-1α silencing groups showed a significantly lower survival fraction than control groups in both T24 and 5637 cells lines (p < 0.05, Figures 3 A and B). The data imply that knockdown of IL-1α decreased the proliferative capacity of cancer cells. To study the mechanism of partial elimination of proliferation, proliferation-related proteins such as Ki-67 and PCNA were measured by western blot. Western blot revealed that Ki-67 and PCNA protein were significantly down-regulated in both T24 and 5637 IL-1α silencing cell lines, respectively, as compared to the control group (p < 0.05, Figures 3 C and D). In a word, these data suggested that knockdown of IL-1α may down-regulate Ki-67 and PCNA proteins to reduce BCa cell proliferation.
Figure 3
Role of interleukin 1α (IL-1α) in cell proliferation ability in T24 and 5637 cell lines. A – Colony formation assay. Results showed that stable knockdown of IL-1α by the adeno-associated virus in T24 and 5637 cells significantly reduced the ability to proliferate in vitro compared to control cells (p < 0.05). B – MTT proliferation assay. OD values were observed to be decreased in the IL-1α silencing group as compared to control (p < 0.05). C – Western blotting showed that the Ki-67 and proliferating cell nuclear antigen were significantly reduced in the IL-1α silencing group compared to control (p < 0.05). D – Western blotting showed that the Ki67 and proliferating cell nuclear antigen were significantly reduced in the IL1a silencing group compared to control (*P < 0.05)
All results were obtained from at least 3 independent experiments.

Knockdown of IL-1α inhibited migration or invasion in vitro by down-regulating metalloproteinase 2 and 9 proteins
To study cell migration changes, in vitro scratch assay was performed. Interleukin 1α silencing cells showed greater scratch space in T24 and 5637 cell lines, respectively, than cells treated with negative control shRNA (both p < 0.05, Figure 4 A). Cell invasion assays were performed to investigate the role of cancer cell invasion in stable knockdown IL-1α cells. Obviously, the invasive ability of IL-1α targeting shRNA treated cells tended to be partially lost compared to negative control cells 48 h later (both p < 0.05, Figure 4 B). In addition, to investigate possible mechanisms for these changes, we examined proteins involved in invasion and metastasis at the protein level, including matrix metalloproteinases (MMP)-2 and -9, by immunoblotting. The data showed that in IL-1α shRNA treated cells, MMP-2 and MMP-9 protein levels were lower than the control (both p < 0.05, Figures 4 C and D). Taken together, the above data suggested that knockdown of IL-1α may inhibit the invasiveness of cancer cells by regulating related proteins.
Figure 4
Role of interleukin 1α (IL-1α) in cell migration and invasion in T24 and 5637 cell lines. A – In vitro scratch assay. Results showed that, compared with control, the IL-1α silencing cells showed bigger scratch space in T24 and 5637 cell lines (p < 0.05). B – Cell invasion assay. Results suggested that the number of invasive cells in the IL1-α6 small hairpin RNA treatment group was significantly lower than that in the control group (p < 0.05). C – Western blot showed that MMP-2 and MMP-9 proteins were significantly decreased in IL1a silencing group compared to control (p < 0.05). D – Western blot showed that MMP-2 and MMP-9 proteins were significantly decreased in IL-1a silencing group compared to control (p < 0.05)
All results were obtained from at least 3 independent experiments.
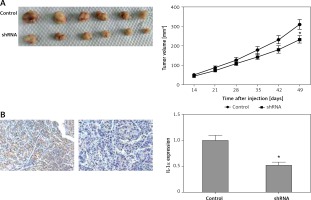
Knockdown of IL-1α inhibited bladder cancer growth in vivo
Based the above results, down-regulated IL-1α significantly inhibited the ability of cell proliferation and invasion or migration in vitro in BCa cell lines. In order to better study the above findings in vivo, xenograft assay was performed. The tumor volume in the down-regulated IL-1α group after 4 weeks management was significantly smaller than that of the control group (p < 0.05, Figure 5 A). To further confirm the silencing effect of IL-1α, immunohistochemical analysis was performed in mouse tumors. The following data showed that our recombinant adeno-associated virus (AAV) significantly reduced IL-1α expression (p < 0.05, Figure 5 B). In summary, the results indicated that knockdown of IL-1α may exert an inhibitory effect in vivo.
Figure 5
Inhibitory role of interleukin 1α (IL-1a) silencing in vivo. A – Xenograft assay. After 4 weeks of treatment, the tumor volume of IL-1a down-regulation group was significantly smaller than control in subcutaneous sites (p < 0.05). B – Results suggested that IL-1a protein level was dramatically reduced by our recombinant the adenoassociated virus in injected tumor xenografts in nude mice by immunohistochemistry assays (p < 0.05)
Discussion
Bladder cancer is the fifth most common malignancy in the world [1]. Despite clinical therapeutics, still 60–70% of patients suffered recurrence, of whom 10–30% eventually had malignant progression, including muscle invasiveness or metastasis [4–6]. The prognosis is usually poor for patients with metastatic disease. In mouse and human genetic studies, IL-1α has been linked to intrinsic and extrinsic pathways [16]. Interleukin 1α has been demonstrated downstream of Ras activation in an oncogenesis model study [17–19]. As an important driver protein, IL-1α activates the downstream genes of the NF-κB pathway and induces the expression of pro-inflammatory cytokines, developing the pre-malignant cells and overt tumors [20–22]. Multiple studies have revealed that IL-1α expression is elevated in various human tumors, such as cervical, gastric, pancreatic, colon, lung, head, and neck cancers and multiple myeloma [23–29]. However, to our knowledge, the biological role of IL-1α in BCa remains unclear.
Consistent with the previous results, the present study showed that IL-1α protein expression in BCa tissue was relatively high compared to the normal bladder tissue adjacent to carcinoma, and high expression of IL-1α protein suggested a poor prognosis (short overall survival time and progression-free survival time). Several oncogenes, including Ret proto-oncogene and MYC proto-oncogene, mediate tumor transformation and activate intrinsic inflammatory cytokines to establish a tumor-promoting microenvironment [17, 30, 31]. Thus, IL-1α expression level might indicate whether the disease is progressing. Our clinical data demonstrated that patients with high IL-1α protein expression were associated with higher probability of malignant distant metastasis and vascular invasion compared with the low expression group, consistent with various studies on the function of IL-1α [23–31]. However, an early study reported that low IL-1α expression decreased survival of BCa patients, which was different from our result, and the reasons are perhaps the different kinds of peoples and the districts [32]. Therefore, it is urgent to explore the mechanism. Due to the limited number of clinical cases, no clear clinical correlation between IL-1α expression and other clinicopathological features including age, gender, tumor stage, tumor grade, lymph node metastasis and recurrence were found.
To confirm this clinical observation, we generated IL1α targeting the shRNA by AAV to silence its expression in BCa cell lines. Although the results were different, we could use similar research methods [33–36]. We studied the expression of several metastasis-associated proteins, such as MMP2 and MMP-9, after IL-1α silencing. The results suggested that IL-1α regulated MMP-2 and MMP-9 to affect cancer invasiveness. In pancreatic ductal adenocarcinoma, Vegardel suggested that production of IL-1α by pancreatic cancer cells induces changes in MMPs and up-regulated the expression and activation of MMP1 and MMP3 and enhanced the migration of pancreatic cancer cells [37]. In addition, there have been many studies showing the potential of IL-1α neutralization in patients with cancer [38–41], which will be our future work to use these antibodies but not this study. Moreover, our data demonstrated that knockdown of IL-1α greatly decreased proliferation by regulating Ki-67 and PCNA proteins in T24 and 5637 cell lines in vitro. In summary, we showed that silencing IL-1α in BCa cell lines may cellular behavior by regulating related proteins. However, the key signaling pathways and molecular mechanisms of IL-1α remain to be elucidated.
We do not know whether IL-1β expression could be affected by IL-1α knockdown and the association between IL-1β and IL-1α. Also combining IL-1α knockdown with other therapies could work especially in immunocompetent mice, and these are our future jobs.
In conclusion, our study demonstrated that IL-1α protein expression in BCa tissue was relatively high compared to the normal bladder tissue adjacent to carcinoma. Furthermore, we showed that IL-1α might be a prognostic biomarker for predicting BCa malignant progression. Functional assays showed that knockdown of IL-1α might inhibit the ability of cancer cells to proliferate and invade or migrate both in vivo and in vitro. Hence, an anti-IL-1α strategy might be a potential form of BCa treatment.