Introduction
Preeclampsia (PE), as a kind of hypertension during pregnancy, is one of the important factors associated with maternal and perinatal death [1]. Studies [2, 3] have shown that among the factors leading to maternal death, PE ranks the third, second only to bleeding and thrombosis. It has been reported [4, 5] that more than 60,000 pregnant women die of PE worldwide every year. PE has serious effects on both the mother and the fetus. If effective treatment is not carried out in time, complications such as pulmonary edema and placental abruption may occur in the mother; complications such as intrauterine growth restriction, premature delivery, respiratory distress syndrome and even death may occur in the fetus [6, 7]. At the same time, PE can also cause serious adverse effects on re-pregnancy; that is, women with a history of PE have 25% to 65% risk of recurrence of PE, 3% risk of placental abruption, and 10% risk of fetal growth restriction [8–10]. Therefore, early diagnosis and effective treatment of PE are essential for the prognosis of patients.
At present, anticoagulant therapy is commonly used clinically. Aspirin and low molecular weight heparin (LMWH) are the most commonly used drugs [11]. The effectiveness and safety of aspirin in the prevention and treatment of PE have been confirmed by a number of studies [12, 13], but there are still controversies regarding the effect and safety of LMWH in the prevention and treatment of PE. There are very few research reports about the applications of aspirin and LMWH in the treatment of PE. Therefore, the effects and safety of aspirin combined with LMWH in the treatment of PE are still controversial. Therefore, this meta-analysis aimed to systematically evaluate the effectiveness and safety of aspirin combined with LMWH in the treatment of PE, and to provide an evidence-based reference for the rational use of PE therapy in clinical practice.
Material and methods
We conducted and presented this meta-analysis in compliance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [14].
Search strategy
We searched the potential related randomized controlled trials (RCTs) with reference to the search strategy formulated by the Cochrane Collaboration. We searched PubMed, EMBASE, Science Direct, Cochrane Central Register of Controlled Trials, China National Knowledge Infrastructure (CNKI), Weipu Database, Wanfang Database, and Chinese Biomedical Literature Database for RCTs on the effects of low-dose aspirin and LMWH in the treatment of PE. The search deadline was January 31, 2021. The search terms included: aspirin, low molecular weight heparin, LMWH, preeclampsia, and the search terms were combined according to the rules of different databases.
The search strategies were jointly formulated by two researchers. According to the established search strategy, the two researchers independently completed the inclusion of the literature, discussed and decided when there were differences, and finally integrated the collected information and data. The research screening process was as follows: preliminary screening of the retrieved documents through the research title and abstract, then further screening through the full text, and finally in strict accordance with the literature inclusion criteria and exclusion criteria to determine the final inclusion of the literature.
Inclusion and exclusion criteria
The inclusion criteria for this meta-analysis were: (1) the patient was diagnosed with PE; (2) RCT study design; (3) the treatment method included the combined use of low-dose aspirin and LMWH; (4) aspirin dose < 150 mg/day; (5) the outcome data could be extracted. The exclusion criteria for this meta-analysis were: (1) animal experimental research, conference abstracts and review articles, retrospective research; (2) studies that lack a control group; (3) articles that were repeatedly published.
Data extraction
Two authors independently evaluated and extracted the data, including: systolic blood pressure, diastolic blood pressure, 24-hour urinary protein, prothrombin time (PT), activated partial thromboplastin time (APTT), fibrinogen (FIB), adverse perinatal outcomes and adverse reactions of pregnant women.
Quality and bias evaluation
We used Cochrane’s risk of bias assessment tool [15] to evaluate the quality and bias of the included RCTs. Seven items were designed to evaluate the risk of bias from the following 6 aspects: (1) sample selection (including random sequence generation and allocation hiding); (2) program implementation (including blinding researchers and subjects); (3) outcome measurement (blind evaluation of study outcome); (4) follow-up (completeness of outcome data); (5) report (selective report of study results); (6) other sources of bias. Each item was rated as “low risk”, “high risk” or “unclear” according to the criteria.
Statistical analysis
We used RevMan5.3 statistical software for meta-analysis. The odds ratio (OR) and its 95% confidence interval (95% CI) were used as the statistics of the combined effect of the count data; the mean difference (MD) and its 95% CI were used as the statistics of the combined effect of the continuous data. The heterogeneity was evaluated by the chi-square test. If the heterogeneity difference between the studies was not statistically significant (p > 0.10 or I 2 < 50%), the combined effect was analyzed by the fixed effects model; otherwise, the random effects model was used for the meta-analysis. In this study, p < 0.05 was considered statistically significant.
Results
Literature search results
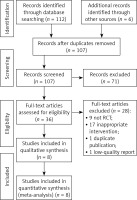
We obtained 118 articles from the initial search. Then we screened out 71 articles by screening titles, abstracts, and duplicate articles. We performed full-text reading of these articles for further screening, and finally included 8 RCTs [16–23]. The study selection process is shown in Figure 1.
Characteristics of included RCTs
Amongst the 8 RCTs included, a total of 861 patients were involved. The basic characteristics of each study are shown in Table I. Generally, the control group received conventional treatment, and the experimental group was treated with low-dose aspirin combined with LMWH on the basis of the treatment of the control group.
Table I
Characteristics of included RCTs
Bias risk assessment
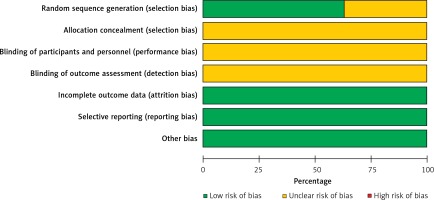
The evaluation results of the Cochrane bias risk assessment tool for 8 included RCTs were as follows: (1) only 5 RCTs [16–18, 22, 23] described specific random sequence generation methods, but the other 3 RCTs [19–21] did not describe specific randomization methods; (2) none of the studies reported specific allocation concealment; (3) none of the studies reported the specific blinding settings; (4) none of the studies described blind evaluation of the outcome; (5) no missing data were found; (6) all pre-set indicators were reported, and no research with reporting bias was found; (7) no other biases were found. The quality and bias evaluation results of the included RCTs are shown in Figures 2 and 3.
Meta-analysis
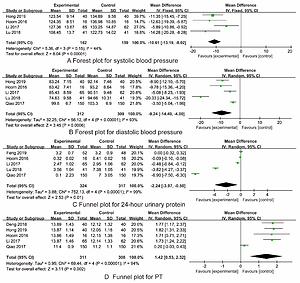
Systolic blood pressure
Four RCTs [16, 19, 21, 23] reported the systolic blood pressure. There was no statistical heterogeneity among the studies (I 2 = 44%, p = 0.15). Therefore, a fixed effect model was used. The synthesized outcome indicated that the difference in systolic blood pressure between the two groups was statistically significant (MD = –10.61, 95% CI: –13.19 – –8.02, p < 0.001) (Figure 4 A).
Diastolic blood pressure
Five RCTs [16–19, 23] reported the diastolic blood pressure. There was statistical heterogeneity among the studies (I2 = 93%, p < 0.001). Therefore, a random effect model was used. The synthesized outcome indicated that the difference in diastolic blood pressure between the two groups was statistically significant (MD = –9.24, 95% CI: –14.49– –4.00, p < 0.001) (Figure 4 B).
24-hour urinary protein
Five RCTs [16, 19, 22] reported the 24-hour urinary protein. There was statistical heterogeneity among the studies (I 2 = 99%, p < 0.001). Therefore, a random effect model was used. The synthesized outcome indicated that the difference in 24-hour urinary protein between the two groups was statistically significant (MD = –2.24, 95% CI: –3.97– –0.50, p = 0.01) (Figure 4 C).
PT
Five RCTs [16, 18–20, 23] reported the PT. There was statistical heterogeneity among the studies (I 2 = 94%, p < 0.001). Therefore, a random effect model was used. The synthesized outcome indicated that the difference in PT between the two groups was statistically significant (MD = 1.42, 95% CI: 0.53–2.32, p = 0.002) (Figure 4 D).
APTT
Five RCTs [16, 18–20, 23] reported the APTT. There was statistical heterogeneity among the studies (I 2 = 56%, p = 0.06). Therefore, a random effect model was used. The synthesized outcome indicated that the difference in APTT between the two groups was statistically significant (MD = 2.91, 95% CI: 2.06–3.75, p < 0.001) (Figure 5 A).
FIB
Four RCTs [16, 19, 20, 23] reported the FIB. There was no statistical heterogeneity among the studies (I 2 = 38%, p = 0.18). Therefore, fixed effect model was used. The synthesized outcome indicated that the difference in FIB between the two groups was statistically significant (MD = –1.24, 95% CI: –1.32– –1.15, p < 0.001) (Figure 5 B).
The adverse perinatal outcomes
Four RCTs [16, 17, 19, 20] reported the adverse perinatal outcomes. There was statistical heterogeneity among the studies (I 2 = 53%, p = 0.009). Therefore, a random effect model was used. The synthesized outcome indicated that the difference in adverse perinatal outcomes between the two groups was statistically significant (MD = 0.41, 95% CI: 0.20–0.85, p = 0.02) (Figure 5 C).
The adverse reactions of pregnant women
Four RCTs [16, 17, 19, 21] reported the adverse reactions of pregnant women. There was no statistical heterogeneity among the studies (I 2 = 0%, p = 0.65). Therefore, a random effect model was used. The synthesized outcome indicated that the difference in the adverse reactions of pregnant women between the two groups was no statistically significant (MD = 0.44, 95% CI: 0.18–1.10, p = 0.08) (Figure 5 D).
Publication bias
The funnel plot on the synthesized outcomes is presented in Figure 6. Even though the funnel plot appeared to be asymmetrical, no publication biases were detected in all the synthesized outcomes (all p > 0.05).
Discussion
It is estimated that 4–5% of pregnant women in the world will suffer from eclampsia [24, 25]. PE brings a great burden in terms of the morbidity and mortality of mothers and babies, and has an important influence on the premature delivery of the fetus and the long-term cardiovascular disease of the mother [26]. The pathophysiological changes of PE include poor trophoblast infiltration, insufficient uterine spiral artery remodeling, and decreased uterine placental perfusion, resulting in clinical manifestations of placental donor insufficiency, leading to insufficient blood supply to the placenta [27, 28]. In addition, this abnormal placenta formation leads to abnormal secretion of anti-angiogenesis and inflammatory proteins, which enter the maternal systemic circulation and damage the maternal systemic blood vessel function [29]. Therefore, early prevention, detection and treatment of PE are of great significance for the prognosis of patients.
Low-dose aspirin is considered the most effective preventive treatment, which can reduce the prevalence of early-onset PE in women. It is generally believed that it is safe for pregnant women to use low-dose aspirin because it will not unintentionally affect the pregnant woman and (or) her unborn fetus [30]. Some researchers [31, 32] believe that the main mechanism of low-dose aspirin is to inhibit platelet aggregation by inhibiting the production of thromboxane. Also, low-dose aspirin has a direct positive effect on the villi trophoblast [33]. However, recent evidence [34] shows that low-dose aspirin prevents the development of preeclampsia by promoting the invasion and migration of trophoblast cells to the uterine artery, interfering with the production of cytokines, and stimulating the pro-angiogenic protein placental growth factor to prevent the development of preeclampsia, thereby inhibiting cell apoptosis and arterial remodeling. Studies [11, 35] have reported that low-dose aspirin taken by people at high risk of PE during gestational weeks 11–14 to 36 gestational weeks can significantly reduce the occurrence of preeclampsia. At the same time, when people with a high incidence of PE start taking aspirin and low molecular weight heparin before 16 weeks of pregnancy, it can effectively prevent PE [36]. The efficacy of aspirin and LMWH in the treatment of patients with preeclampsia remains to be verified. It has been reported that whether aspirin can improve the hypercoagulable state of pregnant women is related to its dose. If the dose is too small, the effect is not obvious. However, excessively large doses can lead to increased adverse reactions such as bleeding in patients [37].
We comprehensively evaluated the efficacy and safety of low-dose (< 150 mg/day) aspirin combined with LMWH in the treatment of PE. The results showed that the blood pressure and 24-hour urine protein in the combined drug use group were significantly lower than in the control group, and its coagulation function (PT, APTT, FIB) was also significantly improved compared with the control group. This suggests that the combination of low-dose aspirin and LMWH has a significant effect in the treatment of PE. The possible reason is that LMWH is a commonly used clinical anticoagulant with anti-inflammatory, anti-immune and protective effects on the vascular endothelium [38]. Its small molecule characteristics make it easy to be absorbed by the body and have a long half-life, and it can also lead to the reduction of platelet aggregation and will ultimately reduce the patient’s thrombosis [39]. Aspirin can inhibit the production of thromboxane, the final product of prostaglandin, by inhibiting cyclooxygenase, which hinders the formation of thrombus, and ultimately improves the clinical symptoms of patients with PE [40]. In addition, studies [41, 42] have shown that the combination therapy improves the soluble vascular endothelial growth factor receptor in patients and reduces the level of ultrasound parameters of placental blood perfusion, and increases the expression of angiogenesis genes in the placenta. This suggests that on the basis of conventional treatment, low-dose aspirin combined with LMWH can improve blood flow in the uterine spiral artery and increase placental blood perfusion. Currently, there are many reports [43, 44] on the active management of labor in women with hypertensive disorder or pre-eclampsia. Based on our findings, the combined use of low-dose aspirin and LMWH may be included in the active management of PE, which still needs further investigation in the future.
This study has the following limitations. Firstly, reports on the effects of aspirin combined with LMWH on PE in foreign countries are inconsistent. Most of the studies included in this meta-analysis are from China, and there is still a lack of high-quality research support from other regions. Secondly, the baseline characteristics of some of the included RCTs are incomplete, such as the lack of descriptions of gestational age and pregnancy details, and the outcome indicators of the included RCTs are not all consistent. Additionally, several studies [45–47] have reported the possible role of miRNAs in PE onset, both as increased or decreased expression in placenta or as maternal serum markers, which may be important outcomes for the evaluation of treatment effects and safety. Future studies focused more on the biological changes are needed. Thirdly, most outcome indicators in this present study are heterogeneous, limited by sample size, so we cannot perform more subgroup analysis. Therefore, the results of this study need to be further verified by a rigorously designed, large-sample, multi-center randomized controlled trial.
In conclusion, the use of low-dose aspirin combined with LMWH in the treatment of PE may be beneficial to improve blood pressure, 24-hour proteinuria, and blood coagulation, and it may reduce the occurrence of adverse reactions of pregnant women without increasing adverse perinatal outcomes. Therefore, the effects and safety of low-dose aspirin combined with LMWH may provide a reference for the clinical treatment of PE. However, it is worth noting that the sample size of this meta-analysis is small, and the conclusions of this study need to be further elucidated in more high-quality studies with further in-depth analysis.