Introduction
With the aging of the population and changes in lifestyle, the prevalence of bone diseases and metabolic diseases continues to increase [1], in which hyperuricemia and osteoporosis are especially significant [2]. Osteoporosis is associated with high oxidative stress levels and weak antioxidant barriers [3, 4]. The increase in osteoclastic activity detected in osteoporosis may be responsible for the increase in reactive oxygen species (ROS) production [3]. Serum uric acid is the end product of purine metabolism, and can be detrimental in many metabolic [5] and cardiovascular disorders [6]. Serum uric acid accounts for approximately 50% of extracellular antioxidant activity [7]. Some recent studies have shown that higher serum uric acid levels are positively correlated with biomarkers of better bone health, leading to speculation about the potential protective role of uric acid against bone loss [8–10]. However, urate crystals promoted osteoclast growth and reduced osteoblast activity, suggesting the globally negative effect of uric acid on bone metabolism [11]. Furthermore, other studies have revealed that hyperuricemic status is correlated with osteoporosis [12–14]. Androgen is known to play a very important role in bone metabolism. Anabolic sex hormones are necessary for bone matrix synthesis, and the loss of androgens with aging in men causes osteoporosis [15]. Furthermore, androgen can maintain the stability of bones [8, 16–18]. However, it remains to be determined whether androgen has an effect on bone and bone metabolism under hyperuricemic status. Therefore, the present study was designed to explore the effect of androgen on bone metabolism in hyperuricemic rats through animal experiments.
Material and methods
Experimental materials
Main reagents
Potassium oxonate was produced by Shandong Zhongke Taidou Chemical Co. Ltd., with a purity of > 98%. Adenine was produced by Shanghai ZZBIO Ltd., with a purity of > 95%. The reagent was dissolved with 0.5% sodium carboxymethyl cellulose (CMC-Na) before use.
Experimental animals
All experimental procedures were performed according to the protocols approved by the Animal Ethical and Welfare Committee of Hebei University (No. IACUC-2017005). The rats were provided by Beijing Vital River Laboratory Animal Technology Co., Ltd. Feeding and management conditions: Rats were kept in a specific-pathogen-free (SPF) animal laboratory, with a room temperature of 20 ±2°C and a relative humidity of 60–70%, and maintained on a 12-hour reverse light-dark cycle with ad libitum access to food and water. The weights of rats were measured once a week, and changes in the body weight of rats were monitored and recorded. Rats were given the corresponding drug dosage according to weight.
Experimental methods
Establishment of the animal model
Forty clean, healthy, male 4-month-old Wistar rats, weighing 280–320 g, were used for the present study. After 7 days of adaptive feeding in the laboratory, these rats were randomly divided into four groups using a random number table (n = 10, each): sham operation group, simple hyperuricemic group, hyperuricemic castration group, and simple castration group.
Establishment of the castrated rat model: Rats in these two castration groups were treated with orchiectomy and epididymis resection, while the fat tissue around the testis in rats in the sham operation group was removed. These rats were defined as controls.
Establishment of the model of hyperuricemic rats [19, 20]: From 10 a.m. every day, rats in the simple hyperuricemic group were given 1.0 g/kg of potassium oxonate for continuous gastric lavage, while rats in the hyperuricemic castration group were treated with intragastric administration of 1.0 g/kg of potassium and a subcutaneous injection of 100 mg/kg of potassium oxonate. Rats in other groups were treated by gavage using an equal volume of distilled water.
Adjustment of modeling methods in rats with hyperuricemia [20]: After the level of uric acid decreased in rats in the simple hyperuricemic group and hyperuricemic castration group, the method for the modeling was altered. Rats in the simple hyperuricemic group were continuously gavaged with 1.5 g/kg of potassium oxonate daily, and subcutaneously injected with 200 mg/kg of ethambutol, while rats in the hyperuricemic castration group were treated with the intragastric administration of 1.0 g/kg of potassium oxonate, a subcutaneous injection of 100 mg/kg of potassium oxonate, and a subcutaneous injection of 200 mg/kg of ethambutol, for a total of 12 weeks.
Blood sampling and testing
Rats were fasted for 12 h before each blood sampling. According to the body weight of rats, each rat was anesthetized via intraperitoneal injection of 3% pentobarbital sodium at a dose of 1.5 ml/kg. Then, 2 ml of blood was sampled from the vena angularis in each rat. Afterwards, these rats were continuously gavaged for 12 weeks.
Drug dosage was adjusted according to the body weight of rats, while the administration time and route were the same for rats in the different groups.
Results
General changes in experimental rats
During the experiment, the body weight of rats in the two hyperuricemic groups initially increased, and subsequently decreased. Furthermore, rats in these two groups had listlessness, dry and yellow hair, and reduced movements, while rats in the control group were smart and active, and had shiny body hair and weight gain. Furthermore, rats in each group were given ad libitum access to food and water during the experiment. During the experiment, four rats died in the simple hyperuricemic group and hyperuricemic castration group.
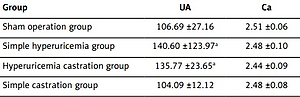
Uric acid and bone metabolism indexes of rats in each group (Tables I–V)
Serum uric acid levels gradually increased at week 4 after the establishment of the model, and the difference was statistically significant (p < 0.05, Table II). During this period, the model of hyperuricemia was successfully established. However, there was no difference in calcium, phosphorus and alkaline phosphatase among all groups, which was possibly due to the short time of the model establishment.
Table I
Uric acid and bone metabolism indexes of rats in each group before the experiment (n = 40, x ± SD, mmol/l)
Table II
Uric acid and bone metabolism indexes of rats in each group at the 4th week (n = 40, x ± SD, mmol/l)
| Group | UA | Ca | P | ALP |
|---|---|---|---|---|
| Sham operation group | 106.69 ±27.16 | 2.51 ±0.06 | 2.37 ±0.05 | 165.34 ±16.17 |
| Simple hyperuricemia group | 140.60 ±123.97a | 2.48 ±0.10 | 2.39 ±0.10 | 159.15 ±27.94 |
| Hyperuricemia castration group | 135.77 ±23.65a | 2.44 ±0.09 | 2.27 ±0.12 | 154.62 ±29.80 |
| Simple castration group | 104.09 ±12.12 | 2.48 ±0.08 | 2.33 ±0.13 | 154.43 ±16.75 |
Table III
Uric acid and bone metabolism indexes of rats in each group at the 6th week (n = 40, x ± SD, mmol/l)
| Group | UA | Ca | P | ALP |
|---|---|---|---|---|
| Sham operation group | 108.91 ±20.16 | 2.40 ±0.13 | 2.33 ±0.38 | 164.11 ±26.78 |
| Simple hyperuricemia group | 136.72 ±22.24a,b | 2.13 ±0.19a,b | 2.33 ±0.23 | 134.50 ±18.79 |
| Hyperuricemia castration group | 135.36 ±24.14b | 2.11 ±0.21a,b | 2.23 ±0.16 | 137.40 ±25.31 |
| Simple castration group | 99.69 ±9.81 | 2.28 ±0.30 | 2.42 ±0.19 | 137.15 ±16.92 |
Table IV
Uric acid and bone metabolism indexes of rats in each group at the 8th week (n = 39, x ± SD, mmol/l)
| Group | UA | Ca | P | ALP |
|---|---|---|---|---|
| Sham operation group | 118.10 ±20.32 | 2.41 ±0.10 | 2.27 ±0.23 | 157.91 ±9.53 |
| Simple hyperuricemia group | 157.22 ±25.38ab | 2.11 ±0.13a | 2.17 ±0.17 | 137.53 ±9.00 |
| Hyperuricemia castration group | 140.15 ±15.00ab | 2.06 ±0.19a | 2.10 ±0.18 | 130.39 ±11.03 |
| Simple castration group | 104.58 ±12.35 | 2.21 ±0.21a | 2.26 ±0.11 | 138.22 ±16.35 |
Table V
Uric acid and bone metabolism indexes of rats in each group at the 12th week (n = 36, x ± SD, mmol/l)
| Group | UA | Ca | P | ALP |
|---|---|---|---|---|
| Sham operation group | 129.41 ±18.83 | 2.41 ±0.17 | 2.22 ±0.19 | 149.18 ±10.44 |
| Simple hyperuricemia group | 172.01 ±21.32a,b | 2.11 ±0.16a | 2.14 ±0.14 | 143.85 ±12.14 |
| Hyperuricemia castration group | 153.83 ±31.87a,b | 2.06 ±0.17a | 1.96 ±0.13a,b,c | 123.16 ±18.10a |
| Simple castration group | 106.59 ±10.71a | 2.21 ±0.22a | 2.26 ±0.20 | 131.15 ±18.17a |
At week 6, uric acid level decreased in the two hyperuricemic groups, and the difference in uric acid levels decreased, and was not statistically significant (p = 0.05).
After week 6, the modeling method was improved. The difference in calcium level in the hyperuricemic castration group was statistically significant, when compared with the castration groups (p = 0.05, Table IV) at week 8. Compared with the castration groups, the difference was statistically significant, indicating that hyperuricemia induced changes in bone metabolism. During this period, the differences in uric acid level among the sham operation group, simple hyperuricemic group and hyperuricemic castration group were statistically significant (p < 0.05). This indicates that the model of hyperuricemia was successful. One rat died in the hyperuricemic castration group, and the cause might be the persistent elevation in uric acid level.
At week 12, the differences in bone metabolism indexes, such as calcium, phosphorus and alkaline phosphatase in rats, were statistically significant (p < 0.05, Table V). During this period, one rat died in the simple hyperuricemic group, while another rat died in the hyperuricemic castration group. In total, four rats died and 36 rats survived, and the reason was considered to be the long-term hyperuricemia in rats. The difference in bone metabolism indexes between the simple hyperuricemic group and hyperuricemic castration group was statistically significant, indicating the effect of androgen on the bone metabolism in hyperuricemic rats.
Discussion
Osteoporosis is a disease characterized by bone fragility and increased risk of fracture [9, 21, 22]. The pathogenesis of osteoporosis is very complex, and involves genetic, lifestyle and environmental factors, as well as other important factors. Androgen plays an important role in bone growth, metabolism, bone mass maintenance and anti-bone loss [23]. In addition, the level of serum uric acid is also affected by androgen. Androgen inhibits uric acid excretion and increases uric acid production [24]. Although uric acid itself can also induce bone damage, the mechanism may be the changes in renal function induced by uric acid crystallization and the rich protein diet taken by hyperuricemic subjects, which leads to a decrease in calcium absorption [25]. In recent years, there has been a view [1] that the occurrence of osteoporosis is correlated with the disorder of differentiation of bone marrow mesenchymal stem cells. That is, bone marrow mesenchymal stem cells excessively differentiate into adipocytes in patients with osteoporosis, which is accompanied by the weakening of osteogenic differentiation ability and bone loss. This is an important cause of osteoporosis. Bone erosion is a common manifestation of chronic tophaceous gout. Monosodium urate monohydrate (MSU) crystals have profound inhibitory effects on osteoblast viability and differentiation, which influences physiological bone turnover, with both excessive osteoclast formation and reduced osteoblast differentiation from mesenchymal stem cells [11].
The present study revealed that at week 4, the difference in uric acid level between the hyper-uricemic groups and the sham operation group was statistically significant, suggesting that the hyperuricemic model of rats was successfully established. However, the differences in blood calcium, phosphorus, alkaline phosphatase and other bone metabolism markers among groups were not statistically significant. These results reveal that hyperuricemia and androgen did not change the bone metabolism in all groups, and this may be due to the short time of the establishment of the model. Hence, the investigators decided to prolong the modeling establishment time.
At week 6, the uric acid level in the hyperuricemic groups also increased, when compared with the sham operation group. But the size of this increase diminished. The level of uric acid decreased in rats in the hyperuricemic groups, when compared with the results in week 4. This may be correlated with the changes in enzymes and the increase in compensatory excretion in rats. Under this condition, the difference in calcium, phosphorus and alkaline phosphatase between these groups was not statistically significant. However, this hyperuricemic status decreased the calcium level of rats at week 6. This confirms the view that hyperuricemia can deteriorate the bone metabolism [16, 26–28]. However, the underlying mechanism remains unknown, at present.
With respect to the correlation between androgen and uric acid, a recent study revealed that the prevalence of hyperuricemia was significantly higher in men than in premenopausal women. That is, androgen increases uric acid levels more effectively than estrogen. The mechanism may be due to the ability of testosterone in androgens to induce the reabsorption of uric acid, allowing the level of uric acid in males to be elevated.
After the adjustment of the modeling protocol, at week 8, the level of uric acid in each group was significantly different from that in the sham operation group. This indicates that the model of hyperuricemia was successful, while the differences in calcium levels between the two castration groups, simple hyperuricemic group and sham operation group were statistically significant. This confirms that androgen has a major effect on bone metabolism in rats.
At week 12, the differences in uric acid levels between the two castration groups and sham operation group were statistically significant. This verifies the effect of androgen on uric acid in non-hyperuricemic rats. That is, androgen increases uric acid levels. This is consistent with a previous research finding [24].
Furthermore, the differences in phosphorus and alkaline phosphatase levels were statistically significant, suggesting the possibility that androgen can decrease bone metabolism in hyperuricemic rats. However, the mechanism of this phenomenon has not been studied in detail. Hence, more experiments are needed to explain the underlying mechanism. Moreover, the present experiment was an animal experiment, and the regulatory mechanism may be different in the inner environment of rats, when compared to humans. Studies with larger sample sizes or further clinical trials are required in the future to explore this mechanism.
In conclusion, androgen can increase serum uric acid levels, and the hyperuricemic status can induce osteoporosis. At the same time, androgen has an effect on bone metabolism under hyperuricemia.