Inappropriate use of antibiotics is one of the main mechanisms in the development of antibiotic resistance. The COVID-19 pandemic may affect antibiotic consumption and lead to increased resistance to antibiotics. Studies show that the use of broad-spectrum antibiotics is common in patients with COVID-19 infection, although confirmed bacterial infection rates are low. It is also reported that adherence to antibiotic management programs has decreased in the fight to save the lives of patients with COVID-19 in this period. The most effective way to combat antimicrobial resistance is to reduce inappropriate antibiotic prescriptions [1–3]. This article aimed to evaluate our hospital’s antibiotic consumption and antibiotic use in the COVID-19 pre-pandemic period (between March 11th, 2019, and March 10th, 2020) and the COVID-19 pandemic period (between March 11th, 2020, and March 10th, 2021) in a tertiary care hospital in the Trakya region of Turkey.
Methods
This single-center retrospective observational study was conducted at Namik Kemal University Hospital, an academic tertiary hospital with 430 beds (including 80 intensive care beds) in Tekirdag, Turkey. In this study, the consumption of antibacterial drugs used intravenously was evaluated 1 year before the beginning of the COVID-19 pandemic and during the year of the pandemic in patients hospitalized at our hospital. The precision of indication-specific antibiotic selection was not questioned. Antibiotic consumption was evaluated with the antibiotic consumption index (ACI). ACI was obtained by dividing the number of defined daily doses (DDD) by 100 days of bed (BD). The study period was divided into two periods: the COVID-19 pre-pandemic period (between March 11th, 2019, and March 10th, 2020) and the COVID-19 pandemic period (between March 11th, 2020, and March 10th, 2021).
This retrospective study was deemed exempt from written informed consent by the Namik Kemal University Hospital review board because it used only retrospective, de-identified patient data.
Results
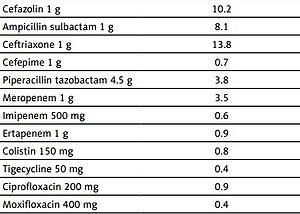
The use of many antibiotics administered intravenously in our hospital increased during the COVID-19 pandemic period compared to the previous year in the Trakya region of Turkey. Antibiotics with the largest increase in ACI value during the pandemic period compared to the previous year increased from 0.4 to 1.8 DDI/100 bed days in moxifloxacin, from 0.9 to 1.9 DDI/100 bed days in ciprofloxacin, from 3.5 to 6.9 DDI/100 bed days in meropenem, from 2.2 to 4.2 DDI/100 bed days in vancomycin, and from 3.8 to 6.4 DDI/100 bed days in piperacillin-tazobactam. The largest decrease in ACI antibiotics was from 0.7 to 0.2 DDI/100 bed days in cefepime, from 0.6 to 0.2 DDI/100 bed days in imipenem, from 8.1 to 3.1 DDI/100 bed days in ampicillin-sulbactam, and from 2.4 to 1.3 DDI/100 bed days in clarithromycin (Table I). A comparison could not be made since the 500 mg parenteral form of azithromycin was administered for the first time in 2020. Teicoplanin, linezolid, and clindamycin were not affected in terms of consumption.
Table I
Consumption of some broad-spectrum antimicrobial agents at Namik Kemal University Hospital during two time periods: COVID-19 pre-pandemic period (between March 11th, 2019, and March 10th, 2020) and COVID-19 pandemic period (between March 11th, 2020, and March 10th, 2021)
Discussion
Bacterial coinfections have played an important role in increased morbidity and mortality during viral respiratory tract infections. Most bacterial respiratory tract infections diagnosed early can be effectively treated with appropriate antibiotics [3]. Bacterial coinfections have been reported in the COVID-19 pandemic as well as in previous pandemics. In a meta-analysis, 7% of patients with COVID-19 were reported to be infected with bacterial pathogens [4, 5]. The most common bacterial co-infections among COVID-19 patients were Mycoplasma pneumonia, Pseudomonas aeruginosa, and Haemophilus influenza. However, there are also controversial data on the coinfection of SARS-CoV-2 with these bacteria. A study by Langford et al. [4] showed that these bacterial pathogens are not common among people with COVID-19. A meta-analysis reported that the rate of Staphylococcus aureus/COVID-19 coinfection was 25.6%.
Studies show that the use of broad-spectrum antibiotics is common in patients with COVID-19 infection, although confirmed bacterial infection rates are low. In many studies, intensive antibiotics are used in hospitalized patients due to COVID-19. In this period, adherence to antibiotic management programs is reported to have decreased in the fight to save the lives of COVID-19 patients. It is stated that most antibiotics in the studies are empirical and that the clinical condition of half of the patients is not serious [1, 2]. The use of many antibiotics in our hospital increased during the pandemic period compared to the previous year.
More than 70% of patients with COVID-19 received some kind of antibiotic with a focus on broad-spectrum agents, such as fluoroquinolones and third-generation cephalosporins. This rate is likely to be higher in patients with severe diseases [4–7]. In different studies, 70% of hospitalized patients were found to receive one or more antibiotics, while almost 100% of hospitalized patients in the ICU received antibiotics [7]. A Bangladeshi study reported that third-generation cephalosporins and meropenem were the most commonly prescribed drugs, while co-amoxiclav and doxycycline were prescribed mainly in Scottish and Singaporean hospitals [8, 9]. At the onset of the pandemic, the combination of third-generation cephalosporins and macrolides was most commonly used in patients with COVID-19. In a study, moxifloxacin, ceftriaxone, and azithromycin were reported to be the antibiotics prescribed to hospitalized patients [10]. In another study, it was found that overall antibiotic consumption in March and April 2020 was not different from the pre-epidemic period, but when considering antibiotics used to treat community-acquired pneumonia (CAP), the average consumption of azithromycin and ceftriaxone was approximately twice that of the pre-epidemic period [11]. In our study, it was found that the antibiotic class with the largest increase in use was quinolones. However, the antibiotics most commonly used were third-generation cephalosporins.
Widespread antibiotic prescribing carries an increased risk of adverse reactions and probably other undesirable effects (such as possible increased bacterial resistance) without benefit. The emergence of antimicrobial-resistant organisms is mainly due to the excessive and inappropriate use of antibiotics. Therefore, optimal antimicrobial use in COVID-19 patients is critical. Many studies have reported an increase in multidrug-resistant organisms (MDRO) during the COVID-19 pandemic. The cause is multifactorial and is related to the high use of antimicrobial agents, especially in patients with COVID-19 with a relatively low rate of co- or secondary infections [2].
In conclusion, in this study, an increase in the consumption of some antibiotics was observed, especially during the pandemic period. Appropriate use of antibiotics is an important strategy in slowing the development of antimicrobial resistance. Studies providing information about antibiotic use will help guide the process.