The presence of EGFR mutations in non-small cell lung cancer (NSCLC) patients is commonly evaluated in tissue material (surgery/biopsy) that may not be both representative of the overall genetic profile, especially for patients with heterogeneous cancer or distant metastases, and sufficient to perform a reliable genetic test (e.g. low tumor cell content). Therefore, a promising alternative in the analysis of EGFR profile is circulating free DNA (cf-DNA) that is released into peripheral blood from both normal and tumor cells that have undergone apoptosis or necrosis [1–5]. Molecular analysis of cf-DNA enables detection and monitoring of EGFR mutations, as well as detection of the acquired resistance for 1st and 2nd generation EGFR-tyrosine kinase inhibitors (TKIs) (erlotinib, gefitinib, afatinib and dacomitinib) caused by Thr790Met substitution that sensitizes NSCLC cells for 3rd generation EGFR-TKIs (osimertinib, rociletinib) [1–3]. EGFR status in plasma or serum is highly concordant with the tumor cells and can be alternatively used in molecular analysis when material from tumor lesions cannot be obtained [1, 4, 5].
In the current study we analyzed the sensitivity of EGFR gene examination in liquid biopsy and utility of this material in monitoring changes in EGFR status during the EGFR-TKI therapy. The studied group included 23 Caucasian patients (8 male and 15 female, median age: 71 ±9 years) with lung adenocarcinoma and known status of activating EGFR mutations (analyzed in formalin-fixed paraffin-embedded (FFPE) and cell blocks). The plasma samples were collected prior to the first EGFR-TKI administration and in 10 patients were re-obtained every 2 months until disease progression. Detailed characteristic of the studied group is presented in Table I and on Figure 1.
Table I
Characteristics of the studied group
The sensitivity of the ctEGFR Mutation Analysis Kit (Entrogen, USA) was 81.2% (9/11) for deletions in exon 19 and 83.33% (10/12) for substitution Leu858Arg in exon 21. Moreover, we did not observe false positive results in control materials. The concordance between plasma and tissue samples reached 82.61% (19/23), which was lower than the concordance demonstrated by Xiong et al. (93.3%), Reck et al. (89%), Douillard et al. (94.3%) and Mok et al. (88%) [1, 2, 4, 5]. On the other hand, Yao et al. observed lower concordance (78.21%) between plasma and tissue samples in next generation sequencing (NGS) analysis [6]. However, nowadays digital droplet PCR (dd-PCR), BEAMing PCR and NGS techniques provide the highest sensitivity for mutation analysis in plasma samples and they are the most recommended [2, 6, 7].
In our study most of the plasma samples showed a low copy number (LCN) content of mutated DNA (mean: 7.44%; range: 0.02–23.8%). The content of mutated DNA in plasma samples was statistically lower than the content of mutated DNA in tissue material (Wilcoxon test: p < 0.000035). We observed a significant positive correlation between concentration of mutated DNA in plasma samples and NSCLC stage (Spearman test: R = +0.53; p = 0.01). Patients with metastatic NSCLC had significantly higher content of mutated cf-DNA than patients with earlier stages of NSCLC (Mann-Whitney U-test: p = 0.02). Zhu et al. suggested the discrepancies between tissue and plasma samples were due to LCN of mutant DNA in plasma [8]. Wei et al. demonstrated that decrease of mutated cf-DNA quantity was statistically significant at the beginning of the treatment, with the exception of patients who carried de novo Thr790Met mutation [9]. Zhu et al. noted decreased concentration of EGFR mutated cf-DNA in plasma during the therapy that significantly correlated with reduction of the tumor burden [10]. Mok et al. reported that continuous presence of EGFR mutations in cf-DNA was a negative predictive factor for overall survival (18.2 months vs. 31.9 months) and correlated with rapid tumor progression [1]. In our study, concentration of mutated cf-DNA dramatic decreased in the first 2 months of treatment and stabilized at a low level. Moreover it did not increase at the moment of clinical progression. Zhou et al. observed that content of mutated cf-DNA was the lowest at the moment of the best response to EGFR-TKIs and increased to the highest level during the progression of disease in some patients, while in the other group of patients the content of mutated cf-DNA did not increase and remained at a stable, low level [3]. Lee et al. showed that NSCLC patients who reached an undetectable level of mutated cf-DNA within the first 2 months of EGFR-TKI treatment had a significantly longer progression free survival (PFS) than patients without reduction of content of mutated cf-DNA (10.1 months vs. 6.3 months) [11]. Similarly, Xiong et al. observed that patients who had a negative result of EGFR analysis in plasma within one month of EGFR-TKI therapy had longer PFS than patients with a positive result of cf-DNA molecular examination (11 months vs. 6 months) [2]. Zhu et al. observed that higher pre-treatment content of mutated cf-DNA shortens PFS during EGFR-TKIs therapy, but reduction of its content to an undetectable level was associated with prolongation of PFS [10].
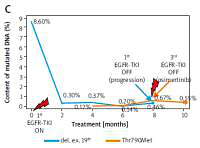
Evaluation of content of cf-DNA with Thr790Met substitution should be performed at the same time as molecular evaluation of tissue biopsy because de novo pre-existing tumor cells with Thr790Met mutation may worsen the prognosis in patients treated with EGFR-TKIs [6]. In our study, the pre-treatment concentration of cf-DNA with Thr790Met substitution was undetectable or at a very low level. Presence of this cf-DNA appeared during the therapy, without increase at the moment of progression. Presence of a low copy number of cf-DNA with a Thr790Met substitution did not affect the outcome of patients treated with 1st and 2nd generation EGFR-TKIs, with the exception of one female patient in whom a steady increase in the mutant DNA content coincided with the clinical progression of the disease. Finally, the patient responded to osimertinib with stabilization of mutant cf-DNA content (Figure 2). Zhu et al. mentioned that the overall incidence of de novo Thr790Met mutation in tumor tissues was 79.9%, with higher frequency in larger tumors [9].
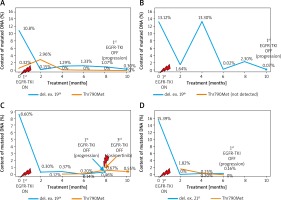
Figure 2
Changes in concentration of mutant DNA in plasma samples during EGFR-TKI treatment in four selected cases (A–D). Rapid decrease of mutated cf-DNA content within first 2 months of EGFR-TKI therapy and stabilization of mutated cf-DNA content at low level within next months of therapy. One patient showed an increase in concentration of mutated cf-DNA in the 4th month of treatment (B) that could be caused by discontinuation and subsequent dose reduction of EGFR-TKI due to toxicity of therapy. When the recommended dose was administered, concentration of mutated cf-DNA reached a low level. The Thr790Met substitution was detectable from the beginning of the therapy or from the second month of this therapy, in the majority of patients. In 1 patient, cf-DNA with Thr790Met substitution was indicated at a detectable level after 4 months of treatment (C). Concentration of this cf-DNA steadily grew, reaching the highest level at the moment of clinical progression of the disease. At that moment, the content of cf-DNA with Thr790Met substitution was higher than the content of cf-DNA with exon 19 deletion. Therefore, the patient received osimertinib, which caused partial remission and stabilization of the disease
Xiong et al. observed dynamic changes in content of cf-DNA with different EGFR mutations in patients treated with 1st and 2nd generation EGFR-TKIs. Among 24 patients who progressed during observation, 15 developed Thr790Met mutation, which was coexistent with an activating mutation, 7 patients lost EGFR gene mutations, 1 patient had only a Thr790Met substitution and 1 patient had only a primary deletion in exon 19. Additionally, in 3 patients presence of Thr790Met substitution was observed without clinical progression [2]. Riediger et al. observed an increase in content of cf-DNA with Thr790Met mutation 2 months prior to clinical progression on EGFR-TKI therapy [12]. In the Zheng et al. study, high content of cf-DNA with Thr790Met mutation was observed 3 months before clinical progression on 1st or 2nd generation EGFR-TKIs and its concentration increased during 4–6 months prior to progression. Moreover, the content of cf-DNA with primary EGFR activating mutations also increased during clinical progression. Additionally, the presence of Thr790Met mutation in plasma was an independent prognostic factor for worse overall survival [13]. Thress et al. and Baijal et al. reported that molecular progression on EGFR- TKI treatment determined by Thr790Met detection in cf-DNA was determined 6 weeks earlier than clinical and radiological progression. Earlier administration of osimertinib according to molecular findings of progression prolonged PFS (10.9 months vs. 5.5 months) and the overall response rate (70% vs. 30%) to 3rd generation EGFR-TKIs in comparison to patients treated with osimertinib after confirmation of progression in radiologic examination [14, 15].
The study was approved by the Ethics Committee of the Medical University of Lublin, Poland (No. KE-0254/131/2011).